
Cool Tool – OCELOT™ System Control Provides Flexible Automation for Process Precision
The biopharma industry has continued to embrace automation as a key part of creating more efficient, cost-effective manufacturing. There are many options for how to automate systems, but a universal requirement is the need for a responsive, built for purpose automation software. In most cases automation software provides a single interface for all applications, while this may seem convenient, it often can’t be configured to meet an end users’ exact process or mode of operation.
OCELOT – Advanced Automation Software
Asahi Kasei Bioprocess America’s (AKBA) OCELOT automation software was designed to be future ready by providing a more customizable solution and to address the shortcomings seen with other platforms. OCELOT is easy to use, universally compatible, and configurable; allowing end users to request exactly how their system will function and look at the time of factory acceptance. The AKBA team can create a tool that fits each process exactly and provide training on using the system with ongoing remote support. OCELOT is designed for easy integration and continued flexibility, providing control even as needs and processes evolve throughout the product life cycle.
In addition to customization, the OCELOT allows end users to view and share data throughout a drug’s journey to market. The ability to view and share this data has become increasingly complex as the products have also increased in complexity, however it is critical for regulatory compliance. The need to collect this data throughout the drug development and manufacturing life cycle has created an avalanche of data that is difficult to manage. Automation is a key tool in managing this data, not only to maintain data integrity but regulatory compliance as well. For this to effectively happen, the system must be able to communicate and collect information from other equipment across the facility. OCELOT provides simplified facility integration and complies with 21 CFR Part 11 by maintaining an audit trail of all actions that occur during or outside of a batch, which can be easily generated into a full report. This report includes any instances of operator intervention, automatic system actions, and even charts and graphs of the recipe and process values.
Downstream Process Automation
The role of automation is particularly important in downstream bioprocessing. Advancements in upstream, such as process intensification, have increased antibody titers dramatically, but in turn has placed more strain on downstream processes, which has caused bottlenecks. Automation can play an important role in eliminating bottlenecks through improved process control and increased productivity and consistency.
The need for smart automation software cuts across many downstream applications. As a result, all automated systems offered by AKBA now include OCELOT, which provides seamless integration with existing plant-wide control systems, allowing far-reaching data collection and analysis. Figure 1 provides a visual representation of facility integration.
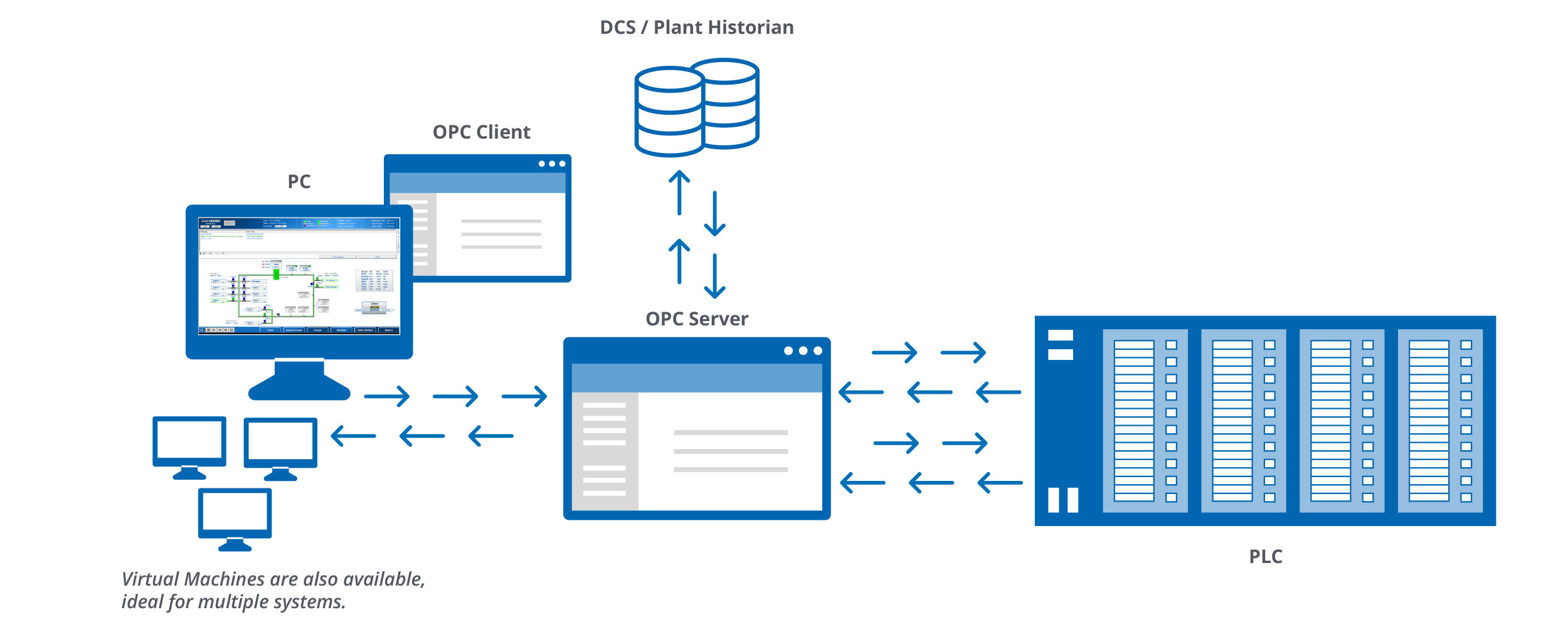
Implementing OCELOT in Key Manufacturing Areas
Recipe Creation and Editing
Recipe creation and editing during drug development and manufacturing is one of the most labor-intensive activities for process engineers. While they are not often the ones executing the recipes they create, they are responsible for monitoring the recipes and making changes as needed. This requires engineers to be present at the equipment every time they want to check in, a cumbersome and time-consuming task. OCELOT eliminates this inefficiency by including a browser-based, remote editing tool through a centralized data server that allows subject matter experts to log into the system from any computer connected to the company intranet. This includes access via a VPN, should a process engineer need or want to work from outside the office ― even at home. They can create new recipes, make and save changes, and even access old batch reports. This functionality can be used simultaneously with other system operations, so users can execute product runs while engineers are preparing the batch for the next set of operations. The user-friendly interface simplifies recipe creation using pre-populated fields and default critical parameters that drive efficiency while minimizing errors and delays. OCELOT’s recipe-editing tool also facilitates scale-up by allowing users to easily validate parameterized recipes and update those parameters at point of use.
Virus Filtration
AKBA’s Planova® Single-Use Virus Filtration Controller (SU-VFC), runs on OCELOT with a kiosk-style interface that walks the user through system operation, selectively presenting only screen or menu options that apply to the current step in the process – mitigating risk of input error along the way. Tube set installation, recipe configuration and batch execution steps are all offered to the operator with simple interactive prompts and pictures. In addition, descriptions of system options, method step types and expected results are crafted by AKBA engineers to appear at point of use. While there are recommended SOPs for each Planova filter type, AKBA understands that individual operations can be slightly different. The AKBA OCELOT platform embraces this marriage of simplicity derived from standard procedure and adaptability to customer specific processes.
Key standard parameters (i.e. Visual Leakage Test setpoints and durations) are automatically set by the system based on the detected filter type from barcode scanning at batch initiation. Flexible parameters are configured out of the box with defaults but can be adjusted as needed by the operator for a variety of filtration approaches – controlling for constant flow, constant pressure, or even constant flux (automatically scaled depending on the installed filtration area).
Inline Buffer Formulation
AKBA’s MOTIV™ Inline Buffer Formulation Systems enable cost-effective, on demand, preparation of large volumes of dilute and pH-conditioned buffers from up to 20X stock concentrates. Each MOTIV is outfitted with OCELOT system control software, which provides intuitive configuration to all inline buffer formulation process steps. Each system can be built to manage an end user’s critical process conditions, with settings, helper text and key process parameters all customized to fit each user’s needs. The OPC-ready architecture plugs into existing DCS, OPC Server or Historian for batch monitoring and control. The OCELOT enables browser-based, remote method configuration and review of batches, even while the system is in use. Thus, permitting upmost flexibility. End users can use recipe configuration, report templates and trend displays to customize daily system operations. The system comes with default parameters, favorite steps, chart presets and report templates for easy repeatability.
Oligonucleotide Synthesis
AKBA’s Cleavage and Deprotection (C&D) System uses OCELOT’s controls to automate post-synthesis column oligonucleotide manufacturing. Thus, eliminating the remaining manual steps in the manufacturing process. The process control screen features a universal run phase for washing, liberation and deprotection steps. There is also a universal wait phase for reaction watching and hold steps. The system also offers optional control of external vessel mixer and external temperature control units.

Key Features and Benefits
- Designed to be simple and straightforward
- OPC-ready architecture plugs into your existing DCS, OPC Server or Historian for batch monitoring and control
- Browser-based to allow remote method configuration and review of batches, even while the system is in use
- Recipe configuration, report templates and trend display allow daily customization of system operations
For more information, please see – fluidmgmt.ak-bio.com/ocelot-system-control/
