
Optimization of a Protein A Chromatography Process for a Herceptin® Biosimilar (Trastuzumab)
As part of our Boston Biotech Week 2016 coverage, we will be writing about some of the posters presented at the conference. One poster that caught my eye for downstream was presented by Oncobiologics and JSR Life Sciences, “Optimization of a Protein A Chromatography Process for a Herceptin® Biosimilar (Trastuzumab).” In the poster, Oncobiologics and JSR Life Sciences describe the steps taken in identifying the most efficient chromatography process.
Poster Highlights Include:
Initial Screening
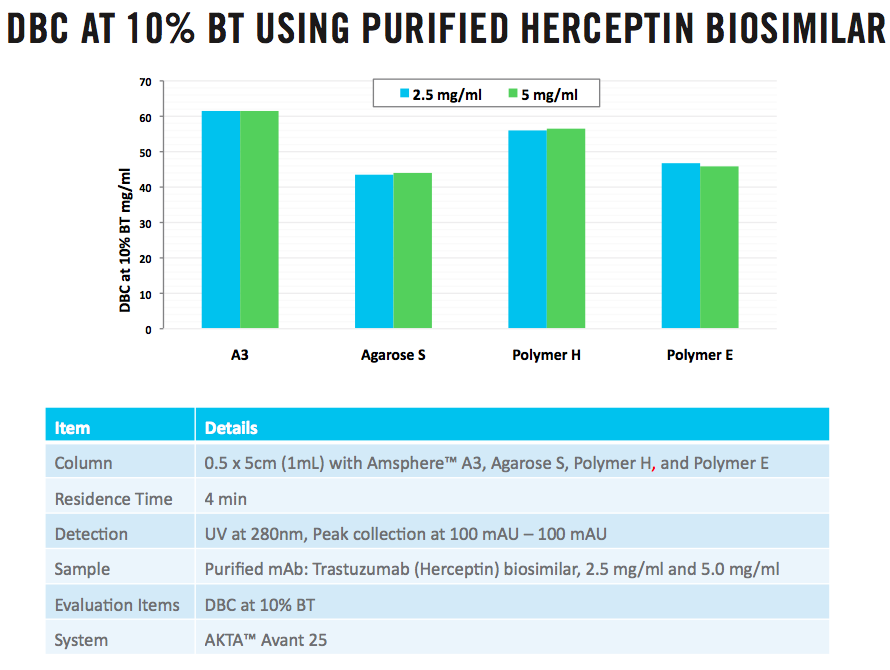
The first step of the study was to screen four Protein A resins from leading industry vendors for use in a capture step, those resins included: Amsphere™ A3, platform resin referred to as Agarose S, and two other polymeric resins referred to as Polymer H and Polymer E. The resins were evaluated at 10% breakthrough utilizing the purified Herceptin biosimilar at two protein concentrations, 2.5 and 5.0 mg/ml and 1 ml columns at 4 minutes residence time. It was observed that substituting Amsphere A3 in the platform capture process led to a 40% increase in dynamic binding capacity relative to two of the resins tested and a 25% dynamic binding increase over the third resin. (Figure 1).
Figure 1:
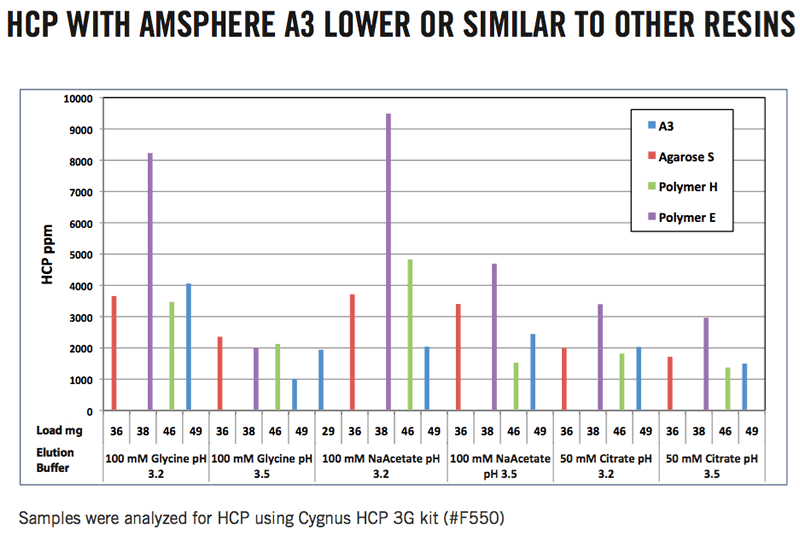
With respect to host cell protein impurity, Amsphere A3 performed as well or better than the other resins (Figure 2).
Figure 2:
Optimization
As a result of the initial screening, Amsphere A3 was selected for further optimization. The elution system and pH were selected after screening studies. The selected elution yielded similar mAb recovery, purity, pH, and eluate volume.
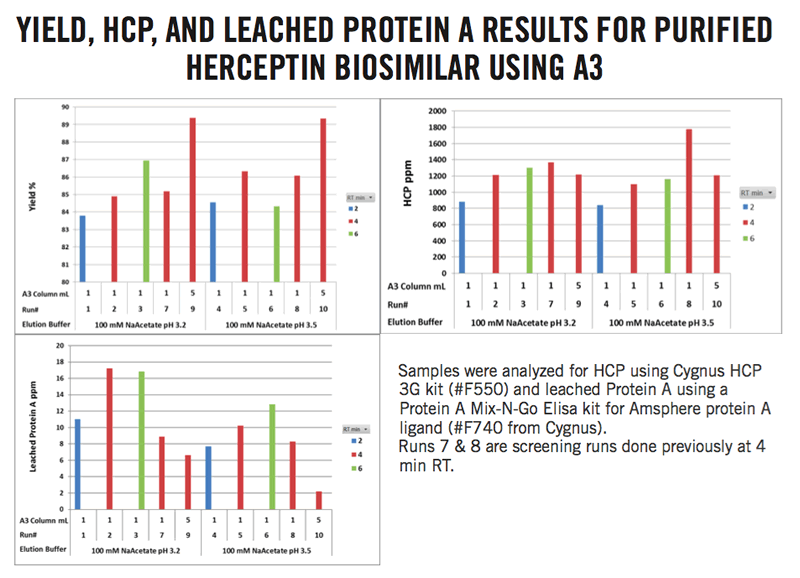
As part of the optimization, the dynamic binding capacity was re-evaluated with Amsphere A3 using a range of residence times bracketing the previously tested 4 minutes. An optimized process utilizing a residence time of 4 minutes or higher with loads of 70-80% of dynamic binding capacity at 10% breakthrough led to increased recoveries and purity of the Herceptin biosimilar. Both host cell protein and leached Protein A impurities were within acceptable ranges. Amsphere A3 had the highest dynamic binding capacity at 10% breakthrough for the Herceptin biosimilar at 4 minutes residence time. (Figure 3).
Figure 3:
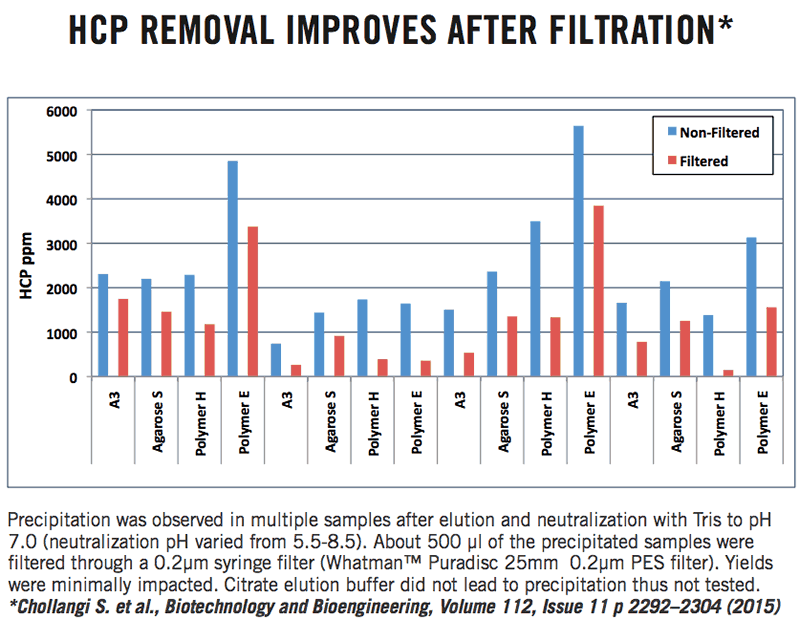
It was also shown that filtration of the eluate after neutralization led to further reduction in HCP (Figure 4).
Figure 4:
Stated Conclusions Based on the Study
- Amsphere A3 had the highest dynamic binding capacity at 10% breakthrough for Herceptin biosimilar at 4 min RT and performed better or similar to other resins at 2 min RT.
- Amsphere A3 performed better in terms of yield and HCP removal.
- An Initial purification process using Amsphere A3 affinity resin for Herceptin biosimilar was developed
- A 4 minute residence time with loading to 70-80% of 10% breakthrough dynamic binding capacity led to high recovery and purity.
- Filtration of eluate after neutralization led to reduction in HCP.
Next steps
Now that the capture step has been developed and optimized with Amsphere A3, next steps include: confirming results at scale with harvest cell culture fluid, demonstrate process reproducibility and assessing product related impurities.
For more data and full study details, please click on the poster below to view in full size.
About Amsphere A3
Amsphere™ A3 is a next generation Protein A chromatography resin for advanced protein separation in downstream processing of therapeutic antibodies manufacturing. Besides an outstanding high capacity, Amsphere™ A3 has an overall improved process robustness, flow characteristics, optimized impurity removal, productivity and resin lifetime.

