Mustang Membrane Technology for AAV Empty and Full Capsid Separation
Interest in Adeno-Associated Virus (AAV) purification approaches have been increasing with recent AAV based gene therapy regulatory approvals. Purification approaches that have been developed for monoclonal antibodies and other recombinant proteins have been modified for the purification of viruses. However, as the demand for gene therapy increases, so does the need for the industry to create a fit-for-purpose purification solution. Resins, which are a mainstay of protein purification, can’t fully address virus purification because of diffusion limitation: The pores are often so small that they exclude viruses, limiting the binding capacity and requiring longer residence times. This, in turn, means operating at lower flow rates and increasing processing times.
Current AAV manufacturing techniques are often characterized as low yield, inefficient, or hard to scale up. These shortcomings result in higher cost of goods and insufficient virus production. Having a platform purification process for AAV, could address many of these challenges, but the use of different AAV serotypes makes achieving this quite difficult.
Empty vs. Full Capsids
In AAV production a mix of capsids, both with and without the DNA payload are generated. The particles with DNA are referred to as full capsids and the ones without are empty. While the impact of empty capsids is not fully understood, they are considered a contaminant and must be removed.
It is common to begin AAV purification with an affinity chromatography step to remove upstream impurities, such as host cell proteins. However, the affinity step is limited because it cannot separate the empty and full capsids. The percentage of empty capsids in upstream can be quite high, in some cases up to 95% of total capsid. Thus, their presence must be controlled to ensure consistent product quality, safety, and efficacy. Since the empty to full capsid ratio cannot be entirely controlled in upstream processing, downstream separation must be part of the downstream purification.
Separating empty from full capsids is very difficult because they have very similar physical properties. They only have a slightly different density and a slightly different charge, (the genetic payload typically confers a slight negative charge to the capsids). Ultracentrifugation can be used to separate based on density. However, ultracentrifugation works at research or small scale, it is not feasible at large or manufacturing scales. Thus, the separation step is frequently the source of process bottlenecks. A chromatographic approach is scalable and; specifically, anion exchange chromatography can utilize the small charge difference to achieve good separation.
Mustang® Membrane Technology
To that end, Pall has added to its Mustang® Q membrane chromatography portfolio sizes specifically fit for gene therapy purification. A recent video “Separation of Empty and Full AAV Capsids,” describes how the Mustang Q technology can be used of efficiently separate empty and full capsids.
In the video, Julio Huato, R&D Engineer at Pall, describes the challenges with AAV purification and the solutions that Mustang® Q membrane technology provides. Mustang® Q is able to reach full AVV 5 capsids and Julio walked through the process development approach to achieving this successful downstream outcome.
During process development, Pall discovered that empty and full capsid separation was not achieved using a linear gradient approach. Instead, a step gradient elution approach with small 1 mS/cm step increments resulted in separation of empty and full capsids (Figure 1).
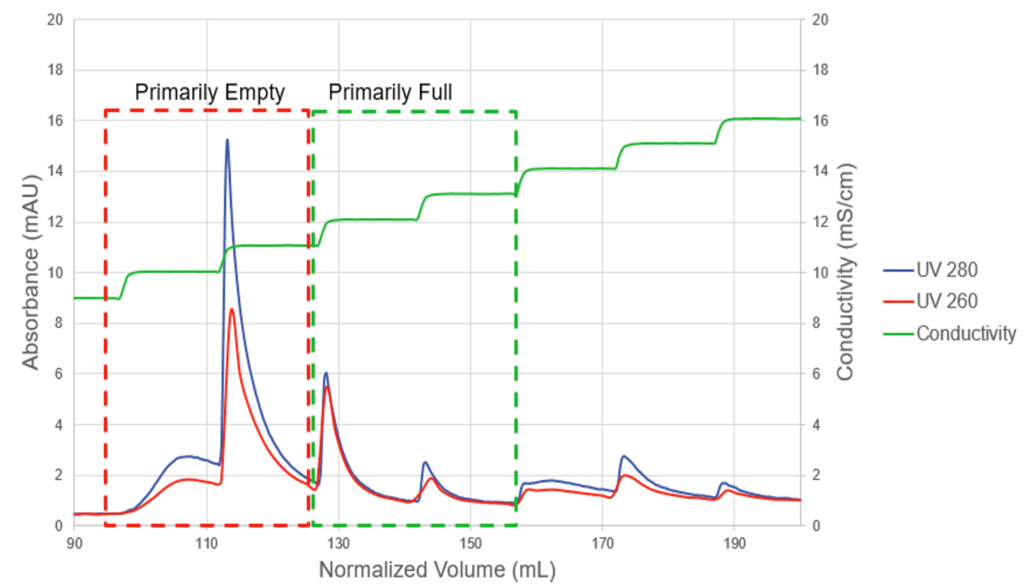 Figure 1. Process Development Tips:
Figure 1. Process Development Tips:
- Use 1 mS/cm step increments, for the step gradient.
- Use of 10-20 MV is recommended for each step.
- A flowrate of up to 10 MV/min is achievable.
The use of 10-20 membrane volumes for each elution step results in a series of discrete elution peaks. The UV absorbance at 260nm and 280nm can provide a rapid visual guide to the purification. Pall recommends this approach as a process development tool. Once separation parameters are identified the elution can be modified to more readily scalable strategy with just two elution steps. Using the Mustang Q membrane approach has resulted in high ratio of full vs. empty capsid (>90%).
In addition, this technology is not diffusion limited, which translates to higher processing flow rates without impacting chromatographic performance or binding capacity. Mustang Q capsules are single-use, ready to use devices that can achieve high viral purity and come in a range of sizes to address different scales (Figure 2).
 Figure 2. Mustang Q Capsule Range
Figure 2. Mustang Q Capsule Range
Mustang Q Capsule Range
- Mustang Q membrane devices are ready to use chromatography adsorbers with bed volumes ranging from 0.86 mL to 5 L.
- Range covers screening to large scale applications.
- These ion exchange supports are polyethersulfone based membranes, with 0.8 µm port size and a cross linked polymeric coating of predominantly quarter nary amine functional groups.
The use of this plug and play technology eliminates the need for column packing, which offers a more efficient and cost-effective manufacturing option and simplifies the downstream operation. All these benefits translate to fast and scalable purification step for AVV production.
Summary
Anion exchange (AEX) chromatography is one of the few methods that can separate empty from full capsids. AEX has been shown to be effective across many AAV serotypes and it could potentially offer a platform purification process for AAV. In addition to their work on AAV 5, Pall has also had success using Mustang Q to purify AAV8 and AAV9. They are seeing comparable results. Pall is working to gather data on all the main AAV serotypes and is actively looking to collaborate with customers on their AAV purification projects. These results make Mustang Q a very attractive candidate for platform AAV empty and full capsid separation.
For more information, please see the video – Separation of Empty and Full AAV Capsids
